 
It is a non-polar covalent molecule with polar bonds. 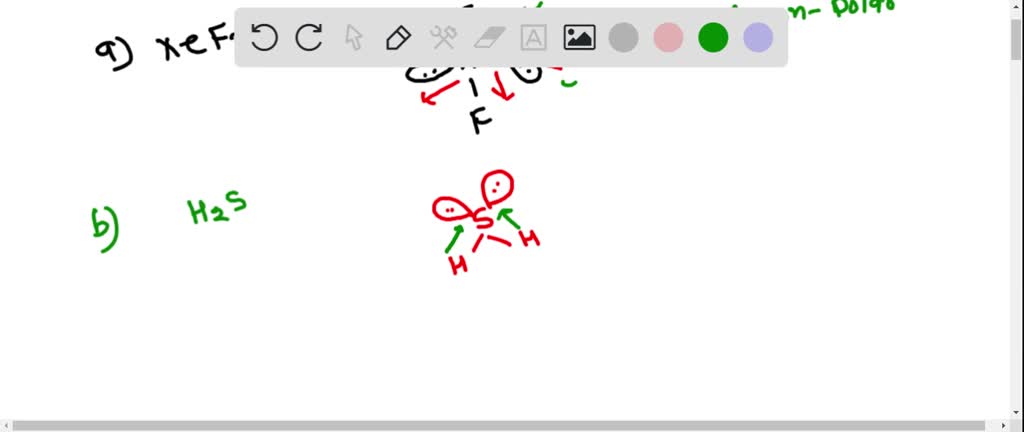
In fact, each of the F atoms is also sp3 hybridized since each has 1 single S-F bond and 3 lone pairs which each count as an electron group. With 3 election groups, it is sp2, and with 4, it is sp3. 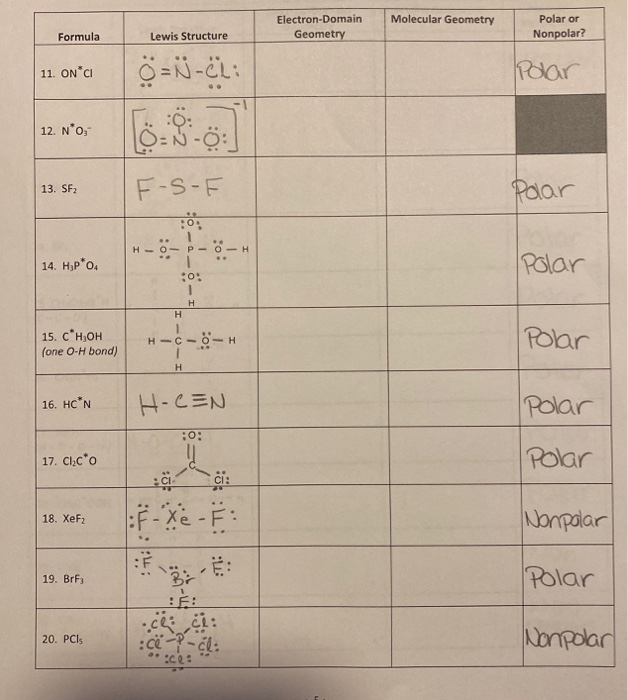
Valence shell theory dictates that with 2 electron groups, the atom is sp hybridized. Hybridization orbitals are determined by the numbers of electron groups present around an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
